Researchers announce positive outcomes from a phase 1 clinical trial that focuses on a vaccine against HIV.
A research study published in the journal of Science on Friday (December 2) outlines positive results from a phase 1 clinical trial performed on humans, which focused on a vaccine against the Human Immunodeficiency Virus (HIV).
As explained by IFLSCIENCE, the results produced from the trial are “a step forward in finding a vaccine against HIV and many other pathogens.”
Titled Vaccination induces HIV broadly neutralizing antibody precursors in humans, the results reported are as follows: “We conducted a first-in-human test of the germline-targeting strategy by evaluating the safety and immune responses of a germline-targeting priming vaccine candidate, eOD-GT8 60mer nanoparticle adjuvanted with AS01B, in the IAVI G001 phase 1 clinical trial.”
It continues, “each participant received two administrations of placebo, low-dose vaccine, or high-dose vaccine 8 weeks apart. The eOD-GT8 immunogen was designed to activate B cell precursors for HIV VRC01-class bnAbs defined by their usage of heavy chain variable gene alleles VH1-2*02 or *04 and any light chain complementarity determining region 3 with a length of five amino acids.”
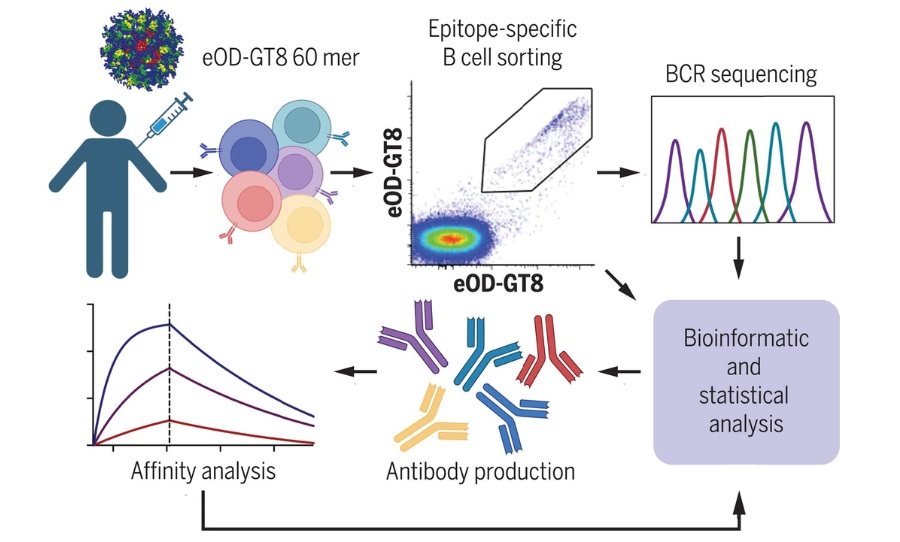
According to the published journal, “the vaccine had a favorable safety profile and induced VRC01-class responses in 97% (35 of 36) of vaccine recipients with median frequencies reaching 0.1% among immunoglobulin G memory B cells in blood. bnAb-precursors shared multiple properties with bnAbs and made substantial gains in somatic hypermutation and affinity with the boost.”
In conclusion, “the results establish clinical proof of concept for the germline-targeting vaccine design priming strategy, support development of boosting regimens to generate VRC01-class bnAb responses against HIV, and encourage application of the germline-targeting strategy to other targets in HIV and other pathogens.”